My baskets
You still don't have anything in your shopping cart
My baskets
You still don't have anything in your shopping cart

{{getOldPrice()}}{{getPrice()}}
 ¡ Buying this product you get {{calculatedProductMenttos()}} menttos !
¡ Buying this product you get {{calculatedProductMenttos()}} menttos !
Without gluten

Without gluten
Milled fruits of Capsicum frutencens Linnaeus. Its color is dark to bright red. Characteristic odor and strongly pungent flavor
Product obtained by grinding the dried, healthy and clean fruits of the varieties of Capsicum frutescens and/or Capsicum Annuum
Its color is dark to bright red. Characteristic odor and strongly pungent flavor
It has a strong pungent flavor
Milled cayenne is the powder resulting from grinding the fruits of one or more species of Capsicum (chili or chili bell pepper), previously dried.
Origin: India/China/Spain/Peru
Capsicum
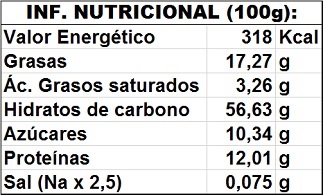
Nutritional value calculated in g/100g:
DECLARATION OF ALLERGENS
According to the data sheet of the supplier(s):
Contains NO GLUTEN AS AN INGREDIENT, BUT NO GUARANTEE OF ABSENCE OF TRACE in packaging other than 1 kg.
ALLERGENS: It does not contain allergens as ingredients
Because of cross-contamination in packaging other than 1 kg may contain Celeriac, nuts, mustard and sesame.
Celeriac, nuts, mustard and sesame
At both our facilities and our supplier's, cleaning and sanitizing processes are carried out after each production to avoid cross-contamination, as well as self-monitoring and verification systems for the confirmation of absence of traces,. However, we cannot claim that the products are free of allergens present in the plant due to accidental contamination
COMMUNITY DIRECTIVE 2006/142/EC ON INGREDIENTS AND LABELLING OF ALLERGENS According to directive 2003/89/EC below is a list of allergenic ingredients that must be mentioned in the labeling for the purpose of consumer protection since the presence of these substances can cause allergies. DECLARATION OF NOT GENETICALLY MODIFIED APPLICABLE LEGISLATION
APTITUDE FOR ETHNIC GROUPS AND OTHERS: HALAL FITNESS : YES, official certificate not available DIABETICS SUITABILITY : YES FIT FOR VEGETARIANS : YES FIT FOR VEGANISTS : YES FIT FOR VEGANS : YES CELIAC SUITABILITY : EXCLUSIVELY IN ORIGINAL FORMAT 1 KG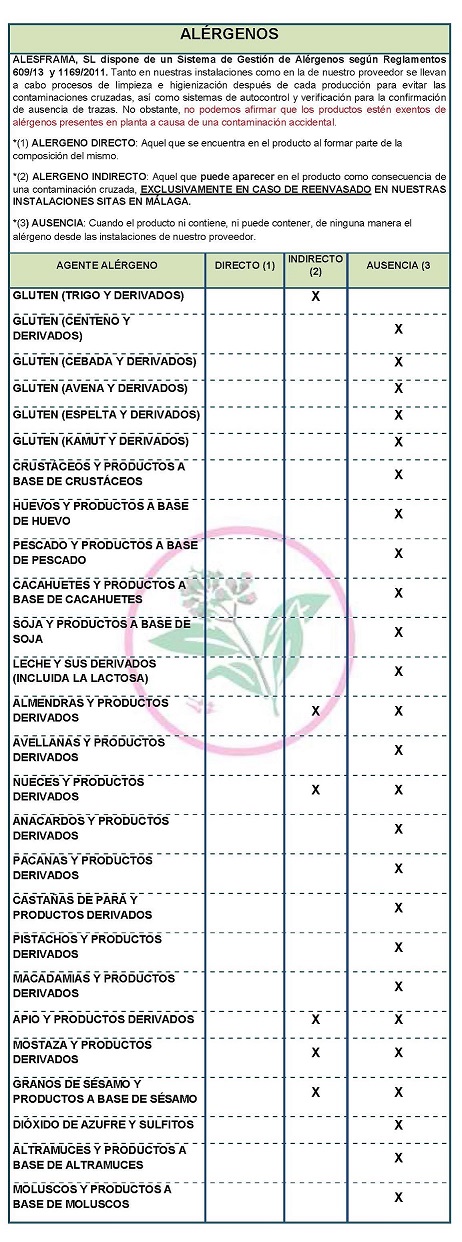
Other non-culinary uses:
Milled fruits of Capsicum frutencens Linnaeus. Its color is dark to bright red. Characteristic odor and strongly pungent flavor
Product obtained by grinding the dried, healthy and clean fruits of the varieties of Capsicum frutescens and/or Capsicum Annuum
Its color is dark to bright red. Characteristic odor and strongly pungent flavor
It has a strong pungent flavor
Milled cayenne is the powder resulting from grinding the fruits of one or more species of Capsicum (chili or chili bell pepper), previously dried.
Origin: India/China/Spain/Peru
Capsicum
Nutritional value calculated in g/100g:

DECLARATION OF ALLERGENS
According to the data sheet of the supplier(s):
Contains NO GLUTEN AS AN INGREDIENT, BUT NO GUARANTEE OF ABSENCE OF TRACE in packaging other than 1 kg.
ALLERGENS: It does not contain allergens as ingredients
Because of cross-contamination in packaging other than 1 kg may contain Celeriac, nuts, mustard and sesame.
Celeriac, nuts, mustard and sesame
At both our facilities and our supplier's, cleaning and sanitizing processes are carried out after each production to avoid cross-contamination, as well as self-monitoring and verification systems for the confirmation of absence of traces,. However, we cannot claim that the products are free of allergens present in the plant due to accidental contamination
COMMUNITY DIRECTIVE 2006/142/EC ON INGREDIENTS AND LABELLING OF ALLERGENS According to directive 2003/89/EC below is a list of allergenic ingredients that must be mentioned in the labeling for the purpose of consumer protection since the presence of these substances can cause allergies. DECLARATION OF NOT GENETICALLY MODIFIED APPLICABLE LEGISLATION
APTITUDE FOR ETHNIC GROUPS AND OTHERS: HALAL FITNESS : YES, official certificate not available DIABETICS SUITABILITY : YES FIT FOR VEGETARIANS : YES FIT FOR VEGANISTS : YES FIT FOR VEGANS : YES CELIAC SUITABILITY : EXCLUSIVELY IN ORIGINAL FORMAT 1 KG
Other non-culinary uses:
